Clearer skin demonstrated in children in AD-1652
(primary endpoint)1-3,a,b
aFull Analysis Set includes all subjects randomized.1
bIn the primary analyses of the efficacy endpoints, subjects who received rescue treatment or with missing data were considered nonresponders.1
cAt Day 1, subjects (baseline weight <30 kg) received 600 mg of DUPIXENT.1
dAt Day 1, subjects (baseline weight ≥30 kg) received 400 mg of DUPIXENT.1
Visible results with DUPIXENT
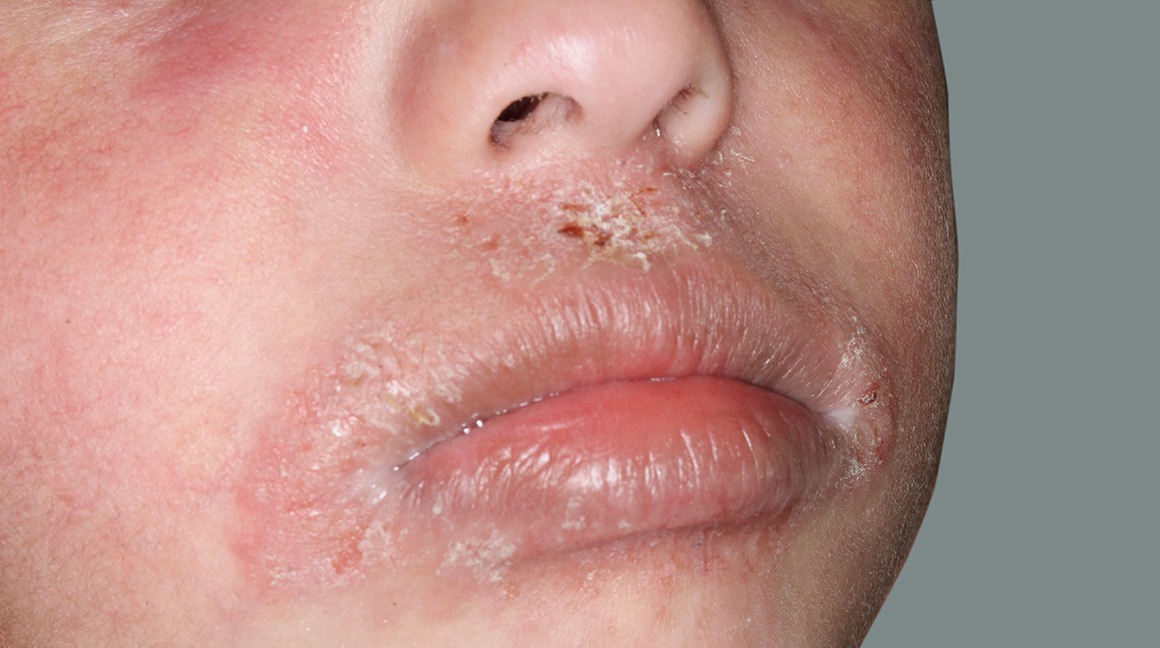
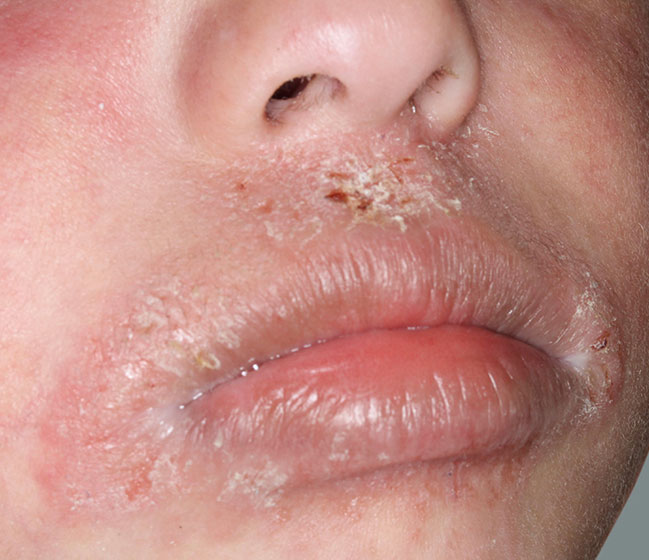
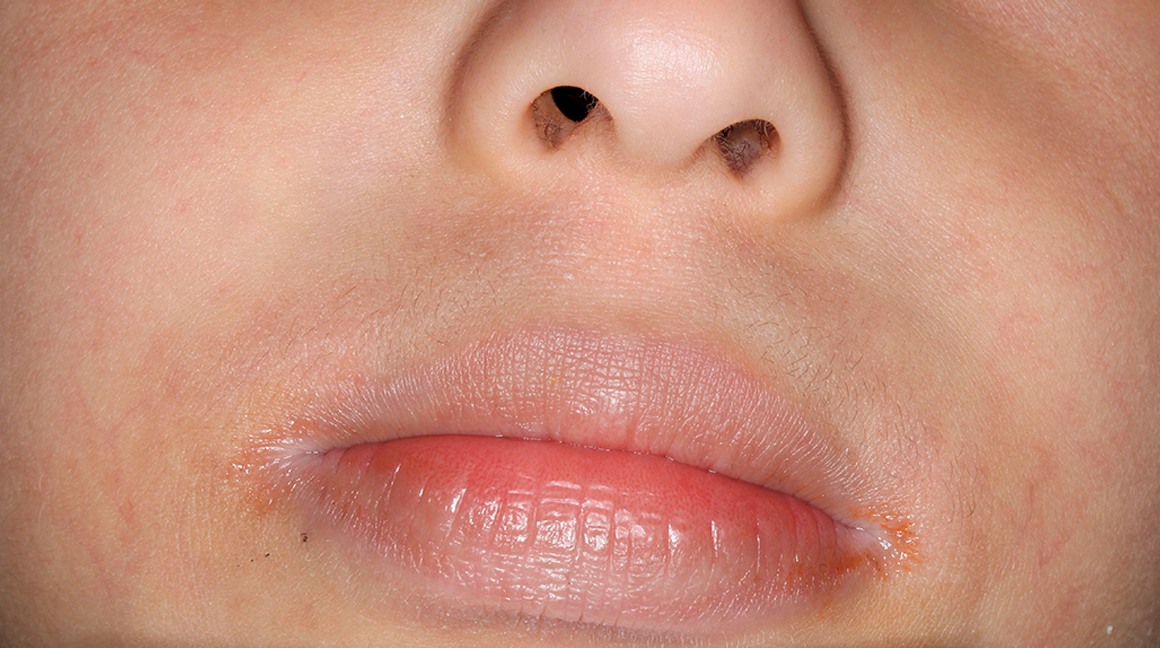
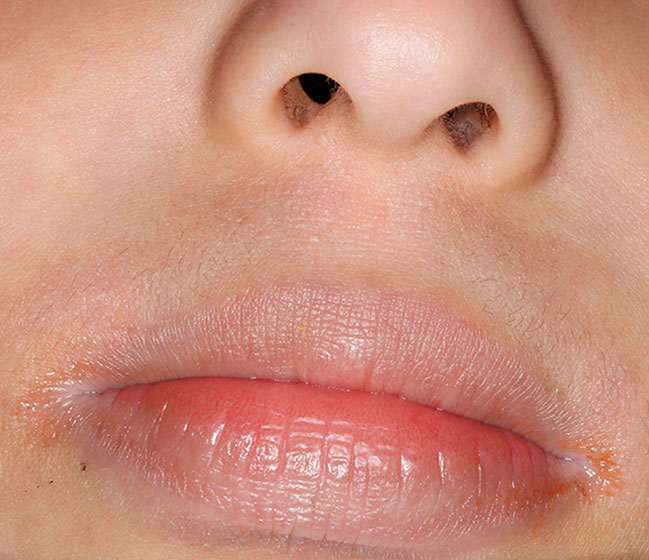
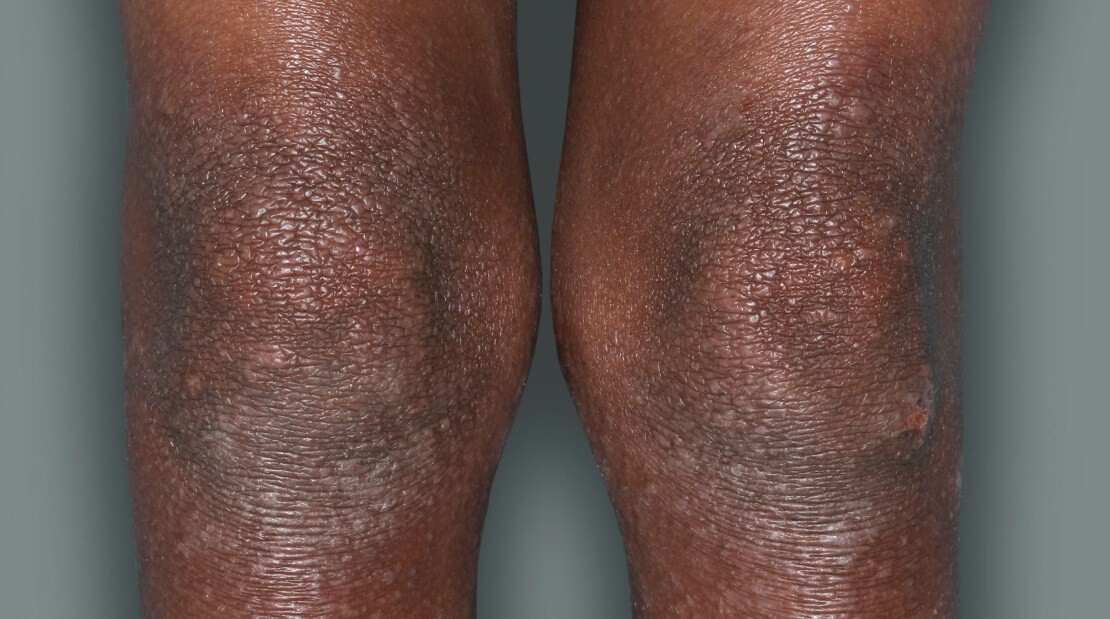
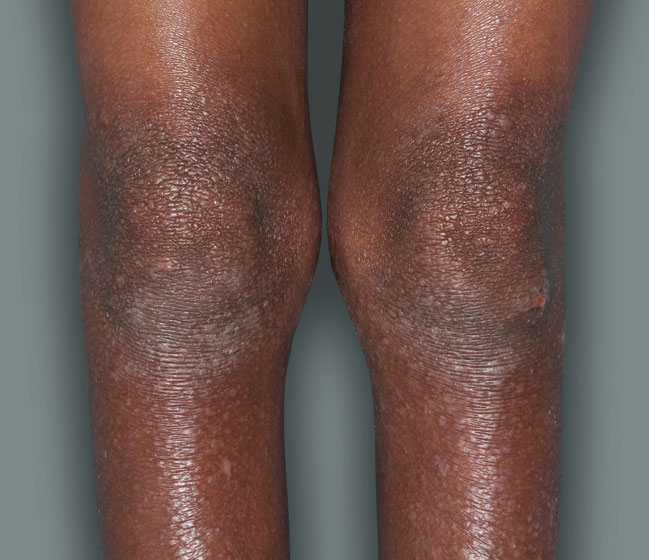
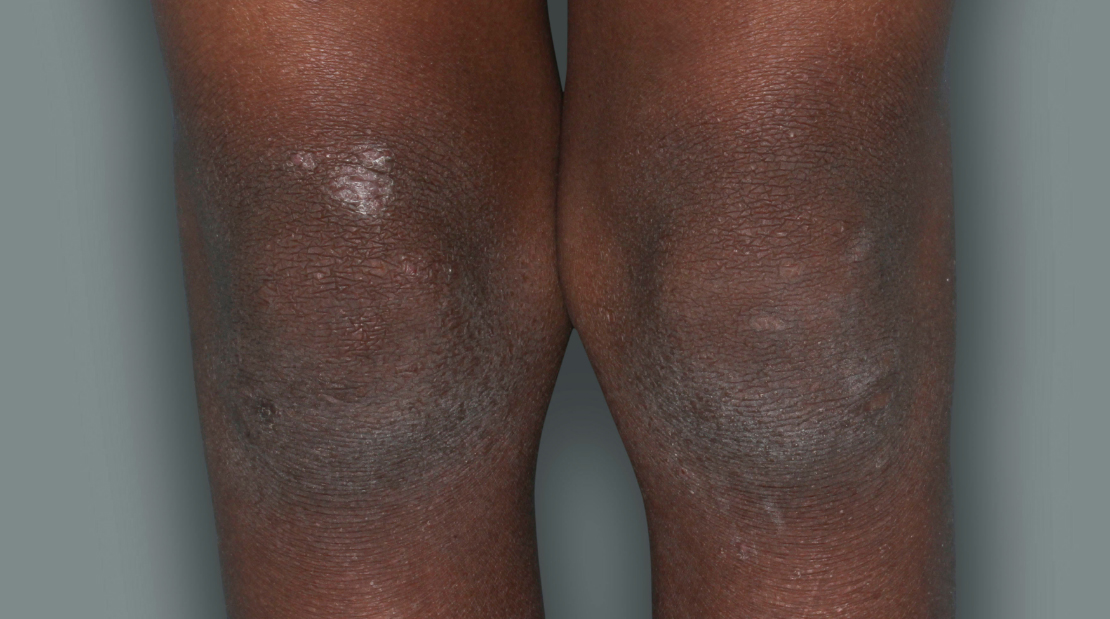
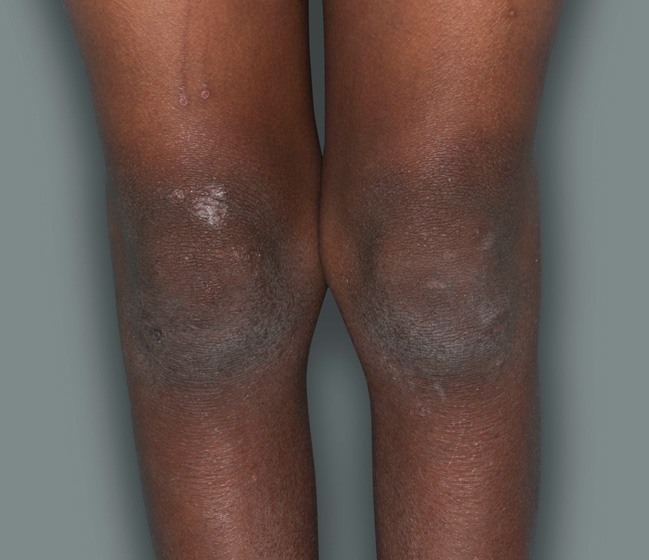
Actual patients in a phase 3 pediatric DUPIXENT trial (AD-1652). All patients were prescribed concomitant TCS based on the clinical trial program. Patients 1, 2, and 3 were considered clinical responders. Individual results may vary.
In DUPIXENT clinical trials including children, the primary endpoint was the proportion of subjects with an Investigator’s Global Assessment (IGA) of 0 (clear) or 1 (almost clear) at Week 16.1
Patient 1: 6-year-old achieved a 3-point improvement in IGA4
RESULTS
Patient 2: 10-year-old achieved a 3-point improvement in IGA4
RESULTS
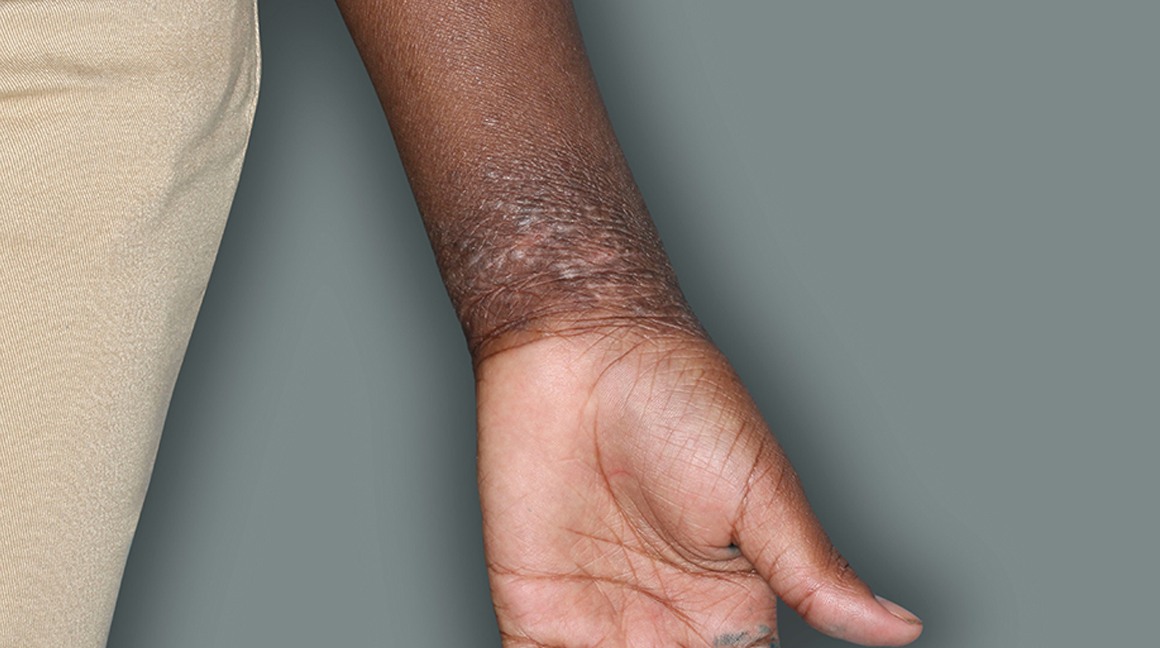
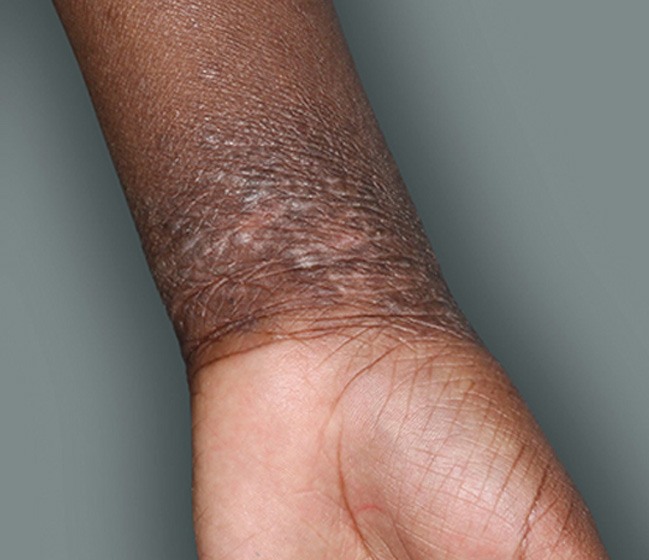
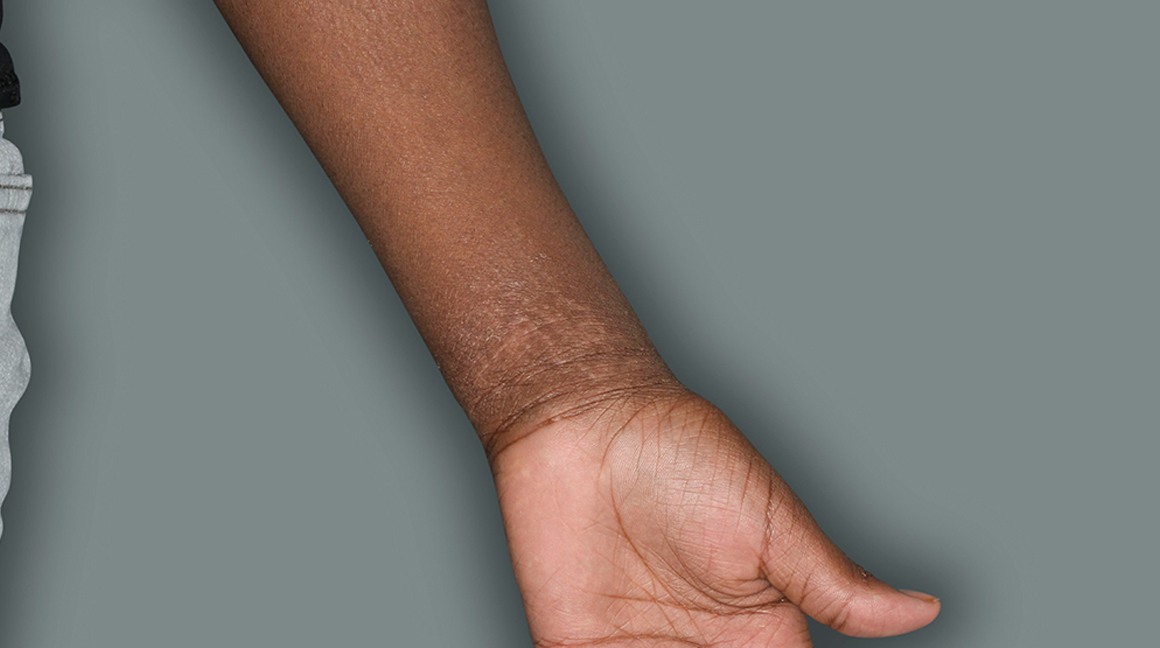
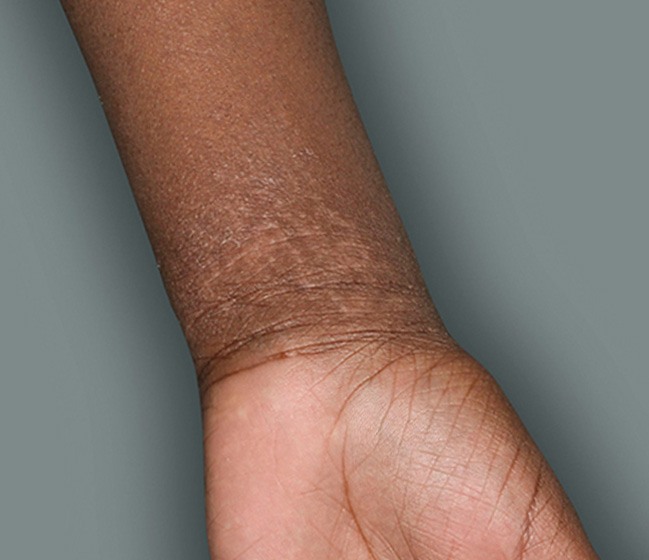
Patient 3: 9-year-old achieved a 3-point improvement in IGA4
RESULTS
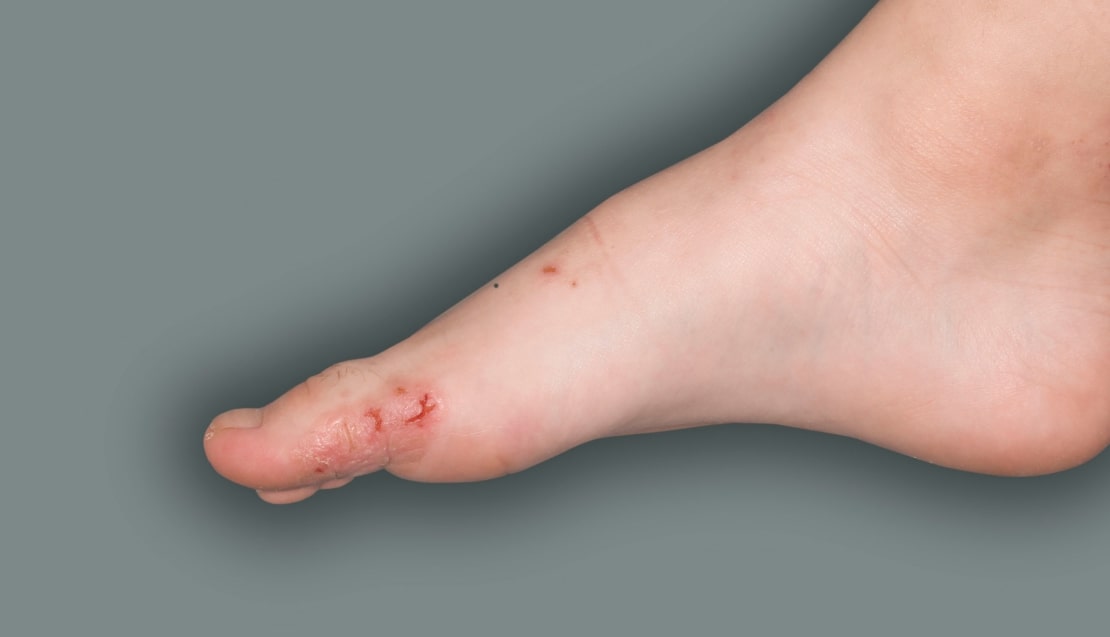
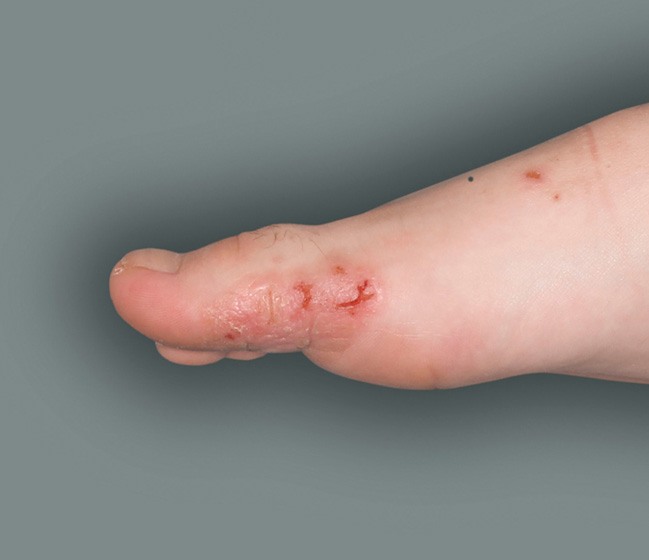
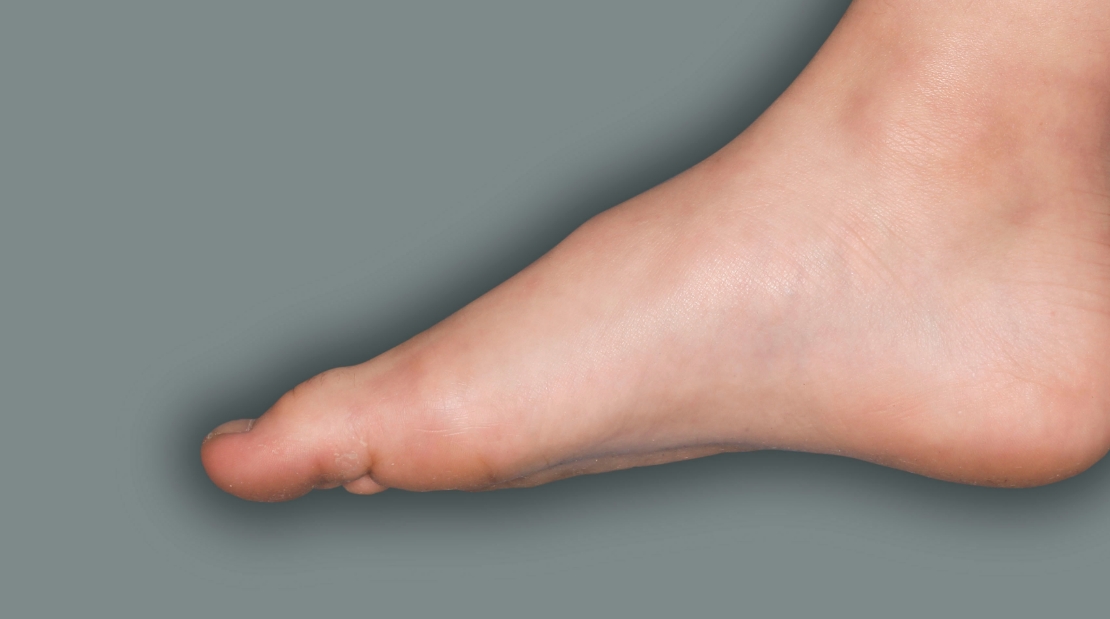
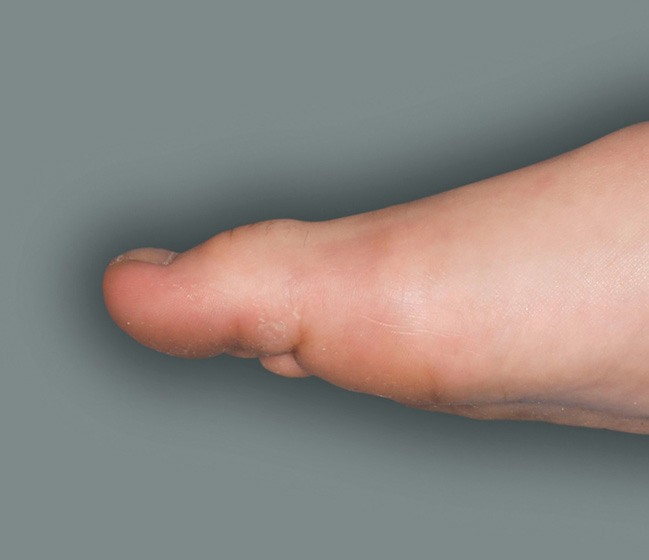
Patient 4: 6-year-old achieved a 1-point improvement in IGA
RESULTS
A clinical responder was defined as a patient achieving IGA 0 or 11
- Patient 4 did not meet the primary endpoint in the clinical trial based on their IGA score at Week 16
Explore more IGA efficacy results
View adult data VIEW ADOLESCENT (12 TO 17 YEARS) DATA VIEW INFANT TO PRESCHOOLER (6 MONTHS TO 5 YEARS) DATA
IGA assesses the overall severity of the clinical signs of atopic dermatitis2
A 0- to 4-point scoring system of the overall severity of atopic dermatitis skin lesions2
Severe
Disease
Severe erythema and severe
papulation/infiltration
Moderate
Disease
Moderate erythema and moderate
papulation/infiltration
Mild
Disease
Mild erythema and mild
papulation/infiltration
Almost
Clear
Just perceptible erythema, and
just perceptible papulation/infiltration
Clear
No inflammatory signs of atopic dermatitis
A clinical responder
was defined as a
patient achieving IGA 0
or 1 and at least a
2-point improvement
from baseline1
IGA, Investigator’s Global Assessment.
Example representation of IGA scoring. Not an actual patient.
READY TO PRESCRIBE DUPIXENT?