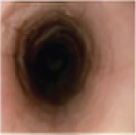
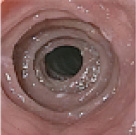
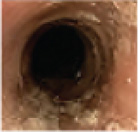
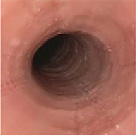
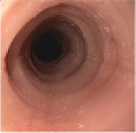
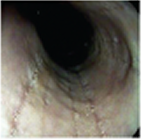
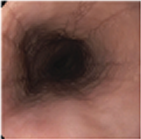
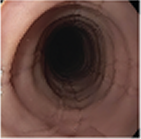
Histologic Remission
- Week 24 (Part A) Coprimary Endpoint: 60% of patients with DUPIXENT (n=42) vs 5% with placebo (n=39) (P<0.001)
- Week 52 (Part A-C) Secondary Endpoint: 56% of patients after 52 weeks of treatment with DUPIXENT (n=34) and 60% after switching to DUPIXENT from placebo at Week 24 (n=30)
)