DUPIXENT SIGNIFICANTLY REDUCED
COPD EXACERBATIONS1-3,a
Annualized rate of moderate or severe COPD exacerbations at Week 52
(primary endpoint)
Number needed
to treat (NNT):
Based on pooled analysis of BOREAS and NOTUS
For every
patients
on DUPIXENT,
moderate or severe
exacerbation was prevented4
The annualized rate of moderate or severe exacerbations for the DUPIXENT and placebo
arms in the pooled analysis was 0.794 and 1.156, respectively (rate ratio 0.687, 95% CI 0.595, 0.793).
Definitive conclusions cannot be reached as the pre-specified pooled analysis was not controlled for multiplicity and the low exacerbation rate observed in the control group might limit the generalizability of our findings.
EXACERBATION REDUCTION DATA
across baseline EOS levels4
of patients with EOS ≥300 cells/μL at screening
- All patients in BOREAS and NOTUS had at least 1 eosinophil count of ≥300 cells/μL during the
4-week screening period- 40% of patients had blood EOS <300 cells/μL at baseline
Annualized rate of moderate or severe COPD exacerbations at Week 52
(post hoc analysis) stratified by baseline blood eosinophil count5,a,*
Definitive conclusions cannot be reached as the pre-specified pooled analysis was not controlled for multiplicity and the low exacerbation rate observed in the control group might limit the generalizability of our findings.
*All patients had at least one eosinophil count ≥300 cells/μL during the 4-week screening, per study criteria.
aModerate exacerbations were defined as exacerbations that resulted in treatment with a systemic glucocorticoid, an antibiotic agent, or both. Severe exacerbations were defined as exacerbations that led to hospitalization or an emergency medical care visit or that resulted in death.1-3
COPD, chronic obstructive pulmonary disease.
DUPIXENT showed improvement in LUNG FUNCTION
Lung function improvement was seen as early as Week 2 and sustained through
Week 52
Post-bronchodilator FEV1
Change in post‑bronchodilator FEV1 from baseline at Week 12 and Week 52 (ITT population; secondary endpoint)1
Pre-bronchodilator FEV1
Significant improvements of similar magnitude were observed in change from baseline in pre-bronchodilator FEV1 at Weeks 12 and 52 in subjects treated with DUPIXENT compared to placebo across the BOREAS and NOTUS trials.1
Patients administered DUPIXENT + triple therapy saw numerical improvement in post‑bronchodilator FEV1 of 134 mL at Week 12 (n=470) and 127 mL at Week 52 (n=362), compared with 67 mL at Week 12 (n=465) and 59 mL at Week 52 (n=359) in patients receiving placebo + triple therapy (LSM change from baseline, ITT population).1
Post-bronchodilator lung function results are descriptive. Definitive conclusions cannot be made.
DUPIXENT IMPROVED PATIENTS’ QUALITY OF LIFE1,2
Improvement as measured by SGRQ6
The sgrq measures the qualitative impact of copd2,6
The SGRQ is a disease-specific instrument designed to measure impact on 3 domains tracking overall health, daily life, and perceived well-being in patients with COPD
of patients reported a clinically
meaningful (≥4-point)
improvement at
Week 52
with DUPIXENT + triple therapy vs 43% for placebo + triple therapy
(N=939; OR: 1.44; 95% CI: 1.10,
1.89; P=0.009)1,2
1.89; P=0.009)1,2
51% responder rate at Week 52
for subjects treated with
DUPIXENT + triple therapy vs 47% for placebo + triple therapy
(N=721;
OR: 1.16; 95% CI: 0.86,
1.58)1,3
NOTUS results are descriptive. Definitive conclusions cannot be made.
DUPIXENT REDUCED
SGRQ SCORESLOWER SGRQ
SCORES
INDICATE IMPROVED
QUALITY OF LIFE1-3
Significant improvement in SGRQ total score at Week 52 IN BOREAS1,2
In NOTUS, patients receiving DUPIXENT + triple therapy (n=362) experienced -9.8
reduction in total SGRQ score vs
-6.4 with placebo + triple therapy (n=359) (LSM
difference: -3.4 [95% CI: -5.8,
-0.9]).1,3
NOTUS results are descriptive. Definitive conclusions cannot be made.
SEE DATA FOR EACH OF THE THREE DOMAINS OF THE SGRQ BELOW*
Symptoms
Cough, sputum
production,
breathlessness, and
wheezing
Activity
Disruption of daily
physical activity
Impacts
Disturbance to
psychosocial function
This domain measures the impact of symptoms like cough, sputum production, breathlessness, and wheezing.6
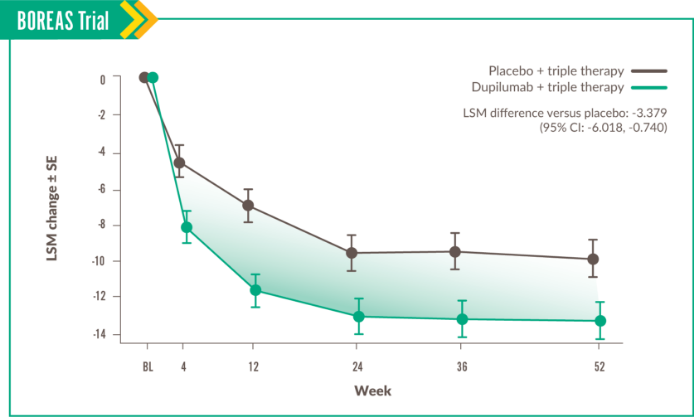
Change in Symptoms score was seen at Week 52, with LSM difference vs placebo of -3.396 (95% CI: -6.397, -0.396).
This domain measures the disruption of daily physical activity.6
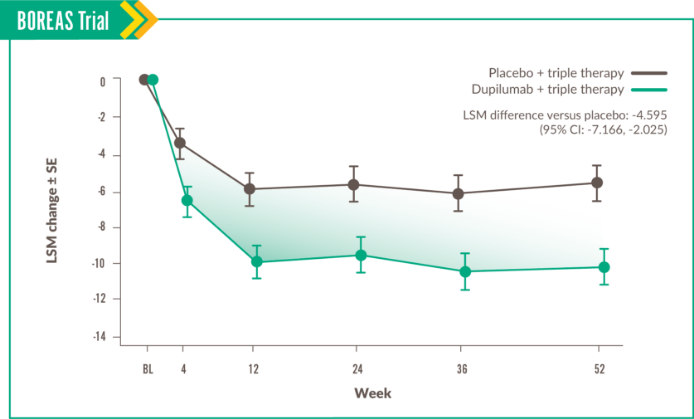
Change in Activity score was seen at Week 52, with LSM difference vs placebo of -3.234 (95% CI: -6.121, -0.348).
This domain measures disturbance to psychosocial function.6
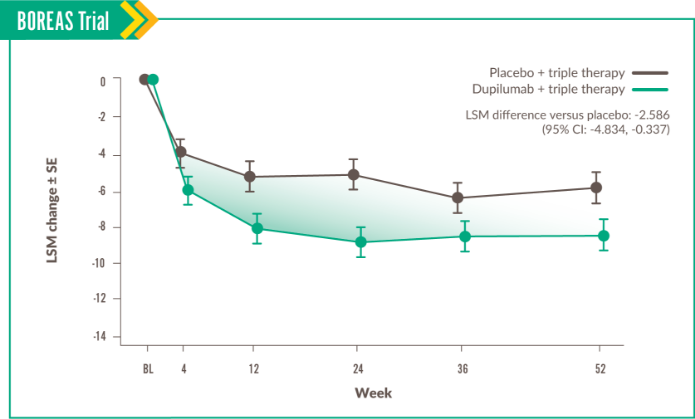
Change in Impacts score was seen at Week 52, with LSM difference vs placebo of -3.261 (95% CI: -6.007, -0.515).
30% reduction in the annualized rate of moderate or severe exacerbationsa for DUPIXENT + triple therapy (0.78 exacerbations per year [n=468] vs 1.10 for placebo + triple therapy [n=471]; P<0.001) (primary endpoint)
34% reduction in the annualized rate of moderate or severe exacerbationsa for DUPIXENT + triple therapy (0.86 exacerbations per year [n=470] vs 1.30 for placebo + triple therapy [n=465]; P<0.001) (primary endpoint)
Based on a post hoc, pooled
analysis of BOREAS and NOTUS
COPD exacerbations leading
to hospital visitsb reduced by
in patients on DUPIXENT + triple therapy
vs placebo + triple therapy
0.16 for placebo (0.12-0.22) vs 0.10 for DUPIXENT (0.07-0.14)
(N= 1874; OR: 0.621; 95% Cl; 0.427 .901) adjusted annualized rate of exacerbations leading to hospital visits9,c
Results are descriptive. Definitive conclusions cannot be made.

bHospital visits is hospitalization or emergency department visits of any duration.
cBased on a post hoc analysis of BOREAS and NOTUS; phase 3, randomized, double-blind, placebo-controlled trials. Adjusted annualized rate of severe exacerbations and/or ED visits of patients ages 40 to 85 years with COPD, moderate-to severe airflow limitation, and type 2 inflammation (screening blood eosinophils ≥300 cells/μL) on LABA/LAMA/ICS. Patients received add-on dupilumab 300 mg or matching placebo Q2W for up to 52 weeks. Severe exacerbations and/or ED visits defined as exacerbations requiring hospitalization or an emergency department visit of any duration.
COPD, chronic obstructive pulmonary disease; ED, emergency department; ICS, inhaled cortiosteroid; LABA, long-acting beta agonist; LAMA, long-acting muscarinic antagonist; OR, odds ratio; Q2W, once every 2 weeks.
Explore study designs and safety data
Learn more about dosage and administration options for patients on DUPIXENT
FREQUENTLY ASKED QUESTIONS
In BOREAS, a 158 mL numerical improvement from baseline in post‑bronchodilator FEV1 was seen at Week 12 with DUPIXENT SC 300 mg Q2W + triple therapy (n=468) vs 84 mL with placebo + triple therapy (n=471) (ITT population, secondary endpoint).1
In NOTUS, patients experienced a 134 mL numerical improvement in post‑bronchodilator FEV1 lung function with DUPIXENT SC 300 mg Q2W + triple therapy (n=470) vs 67 mL with placebo + triple therapy (n=465) at Week 12 (LSM change from baseline, ITT population).1
Post-bronchodilator lung function results are descriptive. Definitive conclusions cannot be made.
Triple therapy consisted of LAMA+LABA+ICS.1
FEV1, forced expiratory volume in 1 second; ICS, inhaled corticosteroid; ITT, intention-to-treat; LABA, long-acting beta agonist; LAMA, long-acting muscarinic antagonist; LSM, least squares mean; Q2W, once every 2 weeks; SC, subcutaneous.
EXPLORE THE LUNG FUNCTIONDATA Discover THE FULL STUDY
DESIGNS
Patients in the BOREAS trial experienced sustained breathing relief at Week 52: a 138 mL improvement from baseline in post‑bronchodilator FEV1 with DUPIXENT SC 300 mg Q2W + triple therapy (n=468) vs 58 mL with placebo + triple therapy (n=471) (ITT population; secondary endpoint).1
Patients in the NOTUS trial experienced sustained breathing relief at Week 52: a 127 mL improvement from baseline in post‑bronchodilator FEV1 with DUPIXENT SC 300 mg Q2W + triple therapy (n=362) vs 59 mL with placebo + triple therapy (n=359) (LSM change from baseline, ITT population).1
Post-bronchodilator lung function results are descriptive. Definitive conclusions cannot be made.
Triple therapy consisted of LAMA+LABA+ICS.1
FEV1, forced expiratory volume in 1 second; ICS, inhaled corticosteroid; ITT, intention-to-treat; LABA, long-acting beta agonist; LAMA, long-acting muscarinic antagonist; Q2W, once every 2 weeks; SC, subcutaneous.
Discover THE FULL STUDYDESIGNS
In the NOTUS trial, the annualized rate of moderate or severe COPD exacerbations declined by 34% in patients receiving DUPIXENT SC 300 mg Q2W + triple therapy (n=470) vs placebo SC Q2W + triple therapy (n=465) over 52 weeks (rate ratio: 0.66 [95% CI: 0.54, 0.82]; P<0.001).1-3
In the BOREAS trial, patients experienced a 30% reduction in the annualized rate of moderate or severe exacerbations at Week 52 with DUPIXENT 300 mg Q2W + triple therapy (n=468) vs placebo SC Q2W + triple therapy (n=471) (rate ratio: 0.71 [95% CI: 0.58, 0.86]; P<0.001).1-3
Triple therapy consisted of LAMA+LABA+ICS.1
Moderate exacerbations were defined as exacerbations that resulted in treatment with a systemic glucocorticoid, an antibiotic agent, or both. Severe exacerbations were defined as exacerbations that led to hospitalization or an emergency medical care visit or that resulted in death.1-3
COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroid; LABA, long-acting beta agonist; LAMA, long-acting muscarinic antagonist; Q2W, once every 2 weeks; SC, subcutaneous.
Discover THE FULL STUDYDESIGNS
In the BOREAS trial, patients receiving DUPIXENT + triple therapy (n=468) saw a 9.7-point improvement in SGRQ total score at Week 52 vs a 6.4-point improvement in patients receiving placebo + triple therapy (n=471; LSM difference: -3.4 [95% CI: -5.5, -1.3]; P=0.002). In NOTUS, patients receiving DUPIXENT + triple therapy (n=362) experienced 9.8-point improvement in total SGRQ score vs 6.4 improvement with placebo + triple therapy (n=359) (LSM difference: -3.4 [95% CI: -5.8, -0.9]).1-3
The BOREAS trial showed a 51% SGRQ responder rate at Week 52 for subjects treated with DUPIXENT vs 43% for placebo (N=939; OR: 1.44; 95% CI: 1.10, 1.89; P=0.009). The NOTUS trial showed a 51% responder rate at Week 52 for subjects treated with DUPIXENT vs 47% for placebo (N=721; OR: 1.16; 95% CI: 0.86, 1.58).1-3
NOTUS results are descriptive. Definitive conclusions cannot be made.
St George’s Respiratory Questionnaire (SGRQ) total score is a patient self-measure of cough, sputum production, breathlessness, and wheezing, as well as disturbances to daily physical activity and psychosocial function. The SGRQ is a 50-item questionnaire designed to measure and quantify health status in adult patients with chronic airflow limitation. Higher score indicates greater disease severity.2,6
COPD, chronic obstructive pulmonary disease; LSM, least squares mean; OR, odds ratio; SGRQ, St George's Respiratory Questionnaire.
EXPLORE ADDITIONAL QUALITY-OF-LIFE DATABased on a post hoc, pooled analysis of BOREAS and NOTUS, COPD exacerbations leading to hospital visitsb reduced by 38% in patients on DUPIXENT + triple therapy vs placebo + triple therapy, 0.16 for placebo (0.12-0.22) vs. 0.10 for DUPIXENT (0.07-0.14) (N=1874; OR 0.621; 95% CI: 0.427, 0.901) adjusted annualized rate of exacerbations leading to hospital visits.9,c
Results are descriptive. Definitive conclusions cannot be made.
bHospital visits is hospitalization or emergency department visits of any duration.
cBased on a post hoc analysis of BOREAS and NOTUS; phase 3, randomized, double-blind, placebo-controlled trials. Adjusted annualized rate of severe exacerbations and/or ED visits of patients ages 40 to 85 years with COPD, moderate-to severe airflow limitation, and type 2 inflammation (screening blood eosinophils ≥300 cells/μL) on LABA/LAMA/ICS. Patients received add-on dupilumab 300 mg or matching placebo Q2W for up to 52 weeks. Severe exacerbations and/or ED visits defined as exacerbations requiring hospitalization or an emergency department visit of any duration.
COPD, chronic obstructive pulmonary disease; ED, emergency department; ICS, inhaled corticosteroid; LABA, long-acting beta agonist; LAMA, long-acting muscarinic antagonist; OR, odds ratio; Q2W, once every 2 weeks.
)