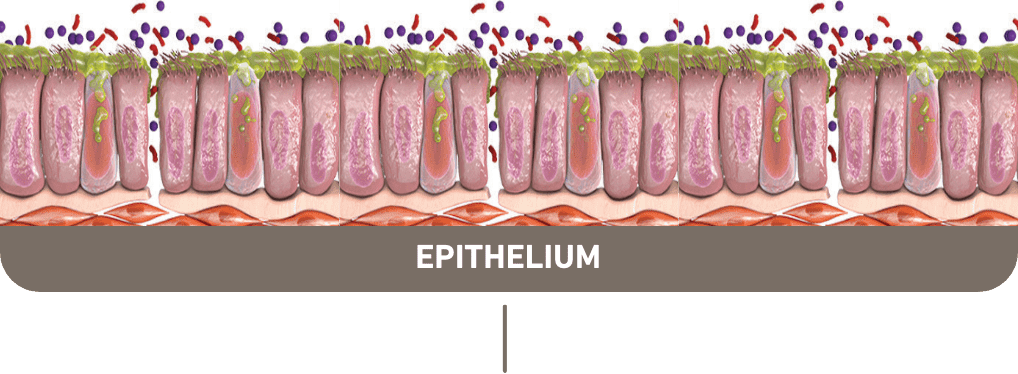
Up to 40% of COPD patients present with type 2 inflammation1-5,a
Multiple manifestations of
type 2 inflammation can
be seen in COPD
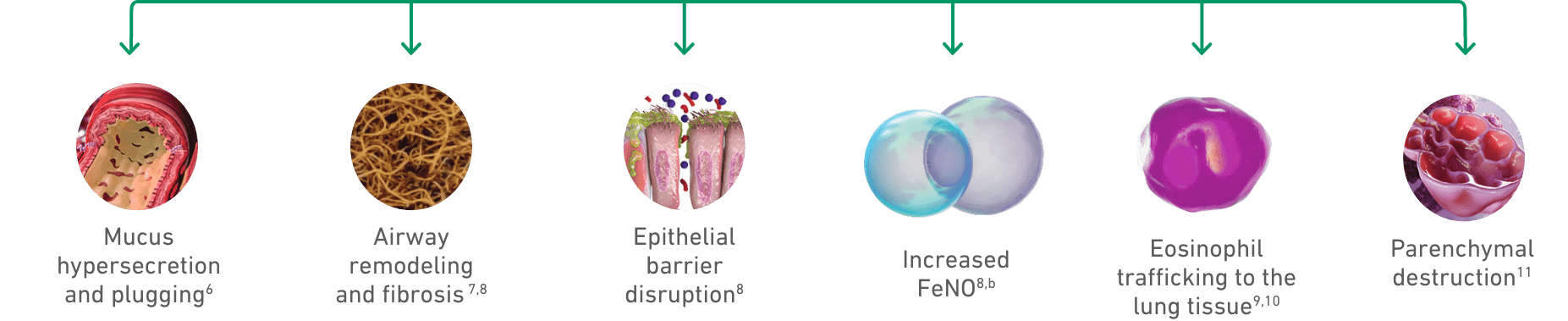
Mucus hypersecretion
Airway remodeling
and fibrosis7,8
Epithelial barrier
disruption8
Increased
FeNO8,b
Eosinophil
trafficking to the
lung tissues9,10
Parenchymal destruction11
How Dupixent Works
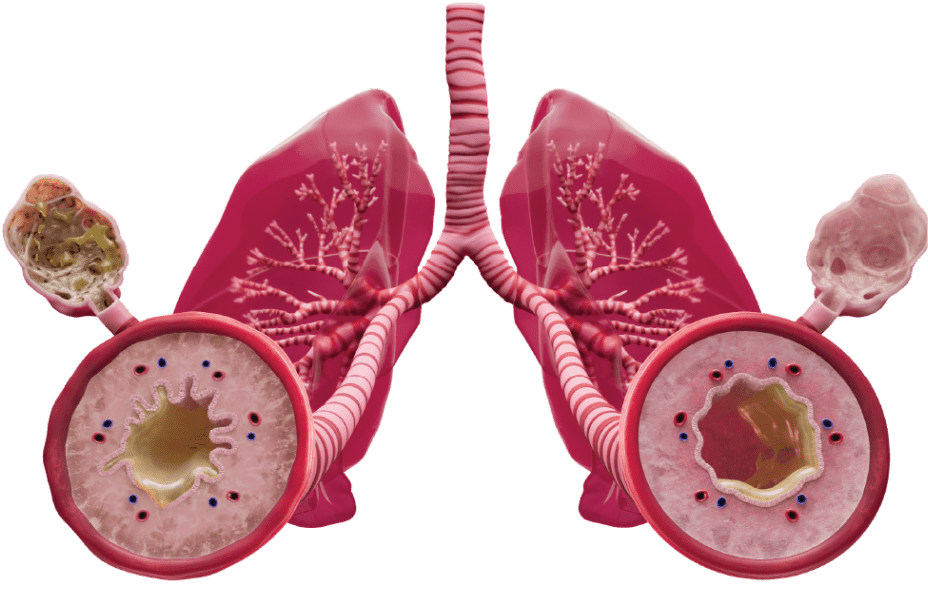
The DUPIXENT Difference DUPIXENT is the only biologic that directly inhibits IL-4 and IL-13 signaling, two of the key drivers of type 2 inflammation12,*
- *The mechanism of dupilumab action has not been definitively established.12
Watch the video below to learn more about the MOA of DUPIXENT
- a Based on findings from 5 studies in COPD patients without asthma. Eosinophil levels used to define type 2 inflammation ranged from ≥300 cells/μL to ≥340 cells/μL (blood), ≥2% in induced sputum, or 3% in peripheral blood. Percentages of patients with type 2 inflammation ranged from 12.3% to ~40%.1-5
- b The role of FeNO in the characterization of COPD is uncertain.
- COPD, chronic obstructive pulmonary disease; FeNO, fractional exhaled nitric oxide.
See which COPD patients may be appropriate for DUPIXENT
FREQUENTLY ASKED QUESTIONS
Approximately 40% of COPD patients present with elevated eosinophils, a biomarker of type 2 inflammation.1-5 Only DUPIXENT directly inhibits IL-4 and IL-13 signaling, two of the key drivers of type 2 inflammation in COPD.12
The mechanism of dupilumab action has not been definitively established.12
DUPIXENT is indicated as an add-on maintenance treatment in adult patients with inadequately controlled COPD and an eosinophilic phenotype.
Limitations of Use: DUPIXENT is not indicated for the relief of acute bronchospasm.
LEARN MORE ABOUT PATIENTS WHO MAY BE AFFECTED BY TYPE 2 INFLAMMATIONIt has been approved across 8 diseases driven in part by Type 2 inflammation and there are >1 million patients on therapy across approved indications worldwide.14,c
DUPIXENT is the first and only dual inhibitor of IL-4 and IL-13—two of the key drivers of type 2 inflammation. The mechanism of dupilumab action has not been definitively established.12
- c The worldwide patient number is largely composed of patients treated with DUPIXENT from 11 countries (Brazil, Canada, China, France, Germany, Italy, Japan, the Netherlands, Spain, UK, and US) and the rest of the world comprising ~12% of this worldwide patient number. This number is composed of the following US approved indications: AD, asthma, COPD, CRSwNP, CSU, PN, and EoE. Data through June 2025.
)