DUPIXENT IMPROVED PATIENTS’ QUALITY OF LIFE1,2
Improvement as measured by SGRQ6
The sgrq measures the qualitative impact of copd2,6
The SGRQ is a disease-specific instrument designed to measure impact on 3 domains tracking overall health, daily life, and perceived well-being in patients with COPD
of patients reported a clinically
meaningful (≥4-point)
improvement at
Week 52
with DUPIXENT + triple therapy vs 43% for placebo + triple therapy
(N=939; OR: 1.44; 95% CI: 1.10,
1.89; P=0.009)1,2
1.89; P=0.009)1,2
51% responder rate at Week 52
for subjects treated with
DUPIXENT + triple therapy vs 47% for placebo + triple therapy
(N=721;
OR: 1.16; 95% CI: 0.86,
1.58)1,3
NOTUS results are descriptive. Definitive conclusions cannot be made.
DUPIXENT REDUCED
SGRQ SCORESLOWER SGRQ
SCORES
INDICATE IMPROVED
QUALITY OF LIFE1-3
Significant improvement in SGRQ total score at Week 52 IN BOREAS1,2
In NOTUS, patients receiving DUPIXENT + triple therapy (n=362) experienced -9.8
reduction in total SGRQ score vs
-6.4 with placebo + triple therapy (n=359) (LSM
difference: -3.4 [95% CI: -5.8,
-0.9]).1,3
NOTUS results are descriptive. Definitive conclusions cannot be made.
SEE DATA FOR EACH OF THE THREE DOMAINS OF THE SGRQ BELOW*
Symptoms
Cough, sputum
production,
breathlessness, and
wheezing
Activity
Disruption of daily
physical activity
Impacts
Disturbance to
psychosocial function
This domain measures the impact of symptoms like cough, sputum production, breathlessness, and wheezing.6
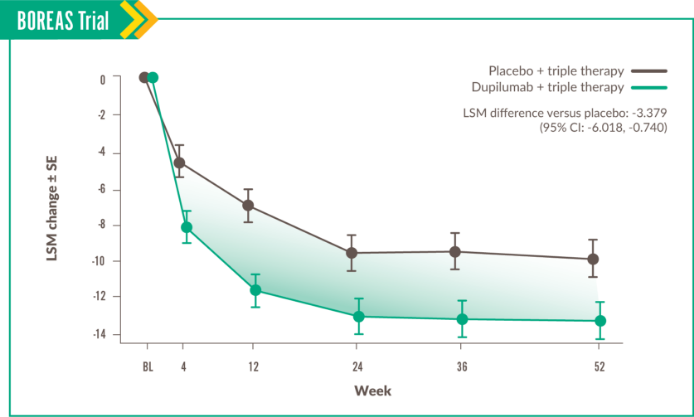
Change in Symptoms score was seen at Week 52, with LSM difference vs placebo of -3.396 (95% CI: -6.397, -0.396).
This domain measures the disruption of daily physical activity.6
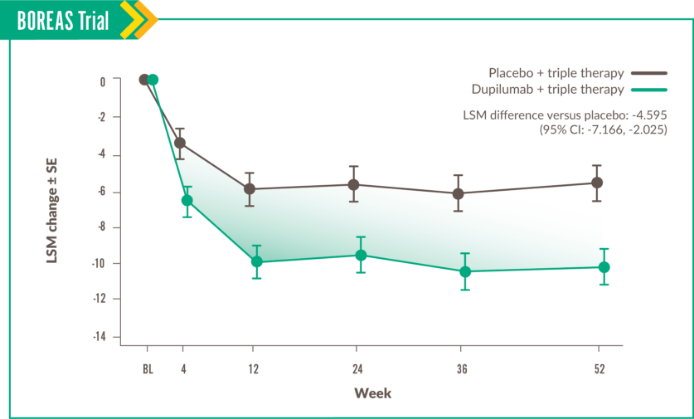
Change in Activity score was seen at Week 52, with LSM difference vs placebo of -3.234 (95% CI: -6.121, -0.348).
This domain measures disturbance to psychosocial function.6
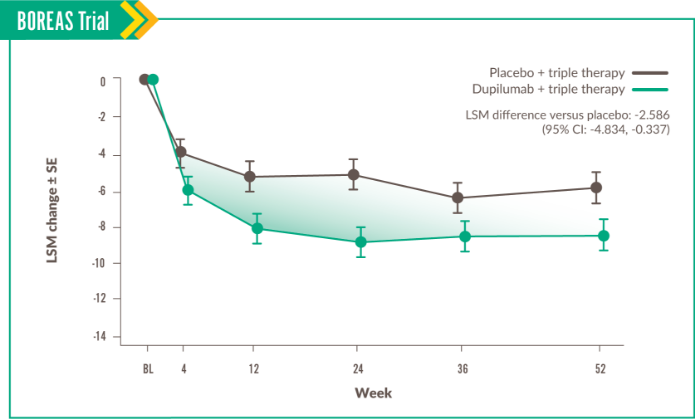
Change in Impacts score was seen at Week 52, with LSM difference vs placebo of -3.261 (95% CI: -6.007, -0.515).